Japan Market Entry for Life Science Tech, MedTech & Diagnostics
We provide a strategic blueprint for innovators in Life Science Tech (AI Platforms, SaaS), MedTech(Medical Devices, SaMD), and Diagnostics (IVD, Reagents) seeking to navigate Japan's complex regulatory and market landscape.
Entering Japan: A huge opportunity, a unique set of challenges
For innovators in Life Science Tech, MedTech, and Diagnostics, Japan is a critical market. But the path to success is unclear.
- Are you facing Japan's unique PMDA / MHLW regulatory hurdles for your medical device, SaMD, or diagnostic product (IVD)?
- Are you struggling to find the right distribution partners or enterprise clients for your specialized technology?
- Do you need to validate if Japanese pharma and academia will adopt your AI platform or SaaS product?
- You need a partner who understands your specific vertical and the local Japanese ecosystem.
Specialized Expertise. Proven Results
- Our Expertise Covers:
- Life Science Tech (AI Drug Discovery, Bioinformatics SaaS, R&D Platforms)
- MedTech (Medical Devices, Software as a Medical Device - SaMD)
- Diagnostics (In-Vitro Diagnostics - IVD, Reagents)
Your Blueprint for a Successful Japan Market Entry
Our Japan Market Entry Assessment is your essential first step. It is a productized consulting service designed to give you maximum strategic value for a fixed price.
| Feature | Basic Assessment | Full Assessment |
| Scope | Market & Go-to-Market Strategy | Market Strategy + Applicable Regulations Research* |
| Price | $3,000 USD | $7,000 USD |
| Delivery | Approx. 4 Weeks | Approx. 6-8 Weeks |
| Payment Terms | 100% Upfront | 50% Upfront / 50% on Completion |
| Best for | Clients in early stages, focused on market viability and strategic direction. | Clients who need a strategic market plan, supported by a foundational list of relevant regulatory documents. |
| Regulatory Checkpoints | Out of Scope | ✔ (Key Component) |
| Deliverable Sample | Download Basic Sample (PDF) | Download Full Sample (PDF) |
*Disclaimer: The Applicable Regulations Research provides a list of publicly available information for reference and does not constitute legal or regulatory advice. The interpretation and completeness of this information should be verified with a qualified professional.
From Blueprint to Market Success: Your Full-Service Partner
The "Japan Market Entry Assessment" is your essential first step. Once your strategic blueprint is complete, we provide hands-on, long-term support to turn your plan into reality. Our ongoing partnership services include:
- Partner Identification & Alliance Building
- Business Development & Market Penetration
- Business Model & Pricing Strategy
- Localization & Market Readiness
These ongoing support services are tailored to your specific needs and are structured as a monthly retainer or project-based fee, discussed after the completion of your initial Assessment.
A Proven Process for Clarity and Confidence
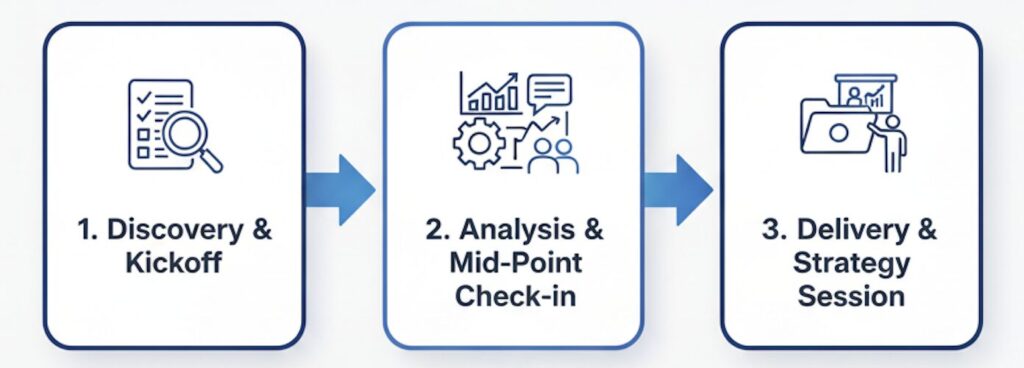
1. Discovery & Kickoff: We start with our questionnaire, followed by a kickoff meeting to align on the "definition of success."
Go to Questionnaire:
2. Analysis & Mid-point Check-in: We conduct our deep-dive analysis and hold a check-in meeting to share early findings and confirm our direction.
3. Delivery & Strategy Session: We deliver the final report and schedule a presentation to walk you through the key findings and answer all your questions.
More Than a Consultant. Your Partner in Japan
Probio is not a large, faceless consulting firm. We are your personal bridge to Japan.
Ready to build your bridge to the Japanese market?
Schedule a complimentary 30-minute call to discuss your goals and see if our service is the right fit for you.
Please note: To ensure the highest quality of service, we accept a limited number of new Assessment projects per quarter.
Have more questions? Read our Frequently Asked Questions (FAQs).
Oversea Partners
InHandPlus
South Korea
A digital health solution that uses AI-powered smartwatches to automatically monitor and record medication intake in real time, enhancing patient engagement and supporting decentralized clinical trials (DCTs).
- Automatic detection and recording of medication intake via camera-equipped smartwatch
- Improved medication adherence and data collection efficiency in DCT settings
- Reminders and real-time interventions to encourage ongoing adherence
- Detection of various medication/measurement behaviors, including injections, inhalers, and blood glucose testing
- Health data visualization and remote support through mobile app and web integration
Extra Horizon
Belgium
A cloud-based Medical Backend-as-a-Service (BaaS) platform with built-in data encryption and regulatory compliance, enabling rapid development of SaMD and remote patient monitoring applications.
- Rapid backend setup for SaMD and connected medical device applications
- End-to-end encrypted infrastructure supporting compliance with GDPR, HIPAA, HDS, DiGA, ISO 27001, and more
- Support for remote patient monitoring and DCT application development
- Advanced healthcare solution development with AI, machine learning, and data analytics
- Accelerated prototyping, regulatory approvals, and global deployment
Laboratoire ODOST
France
Developer and manufacturer of natural and organic-certified cosmetics and personal care products, utilizing thermal spring water from southern France.
- Development and manufacturing of natural formulas with Cosmébio and Ecocert organic certification
- Oral and skin care products made with Castéra-Verduzan thermal spring water
- Custom manufacturing of a wide range of formulations, including gels, foams, sprays, powders, soaps, and aerosols
- Cleanroom manufacturing with strict microbiological and physicochemical quality control
- 100% in-house production in Gers, France, with an environmentally responsible, ethical approach
